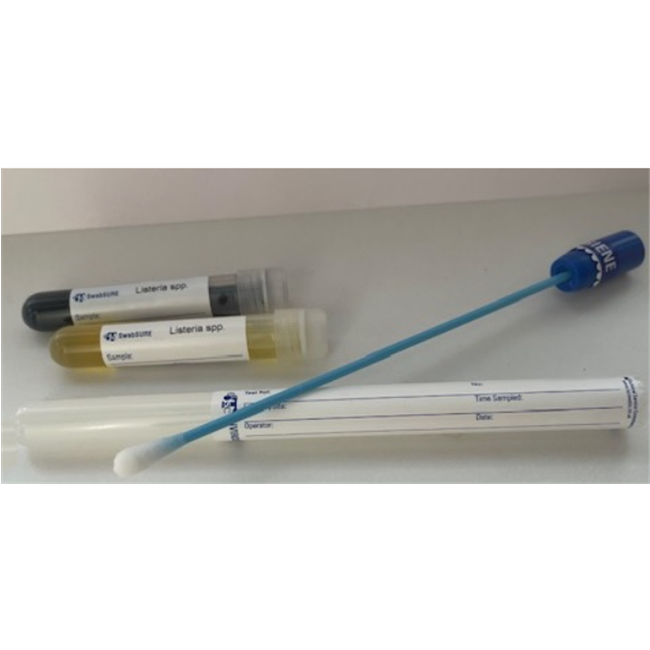
The SwabSURE Listeria swab is a direct test for the
presumptive detection of Listeria spp. including L.
monocytogenes from food contact/preparation areas or
manufacturing environments where regular disinfecting/sanitizing is performed.
Results are easily visualized using the inbuilt colour change system giving
clear results. Kit includes Pre-moistened (in TSC Neutralizing Buffer) 45mm
Breakpoint Swab for simple sample transfer. ISO18593:2018 compliant when used
with a template.
Media Inhibits/suppresses other background microorganisms
such as: Bacillus spp., Enterococcus spp., Micrococcus spp
and Gram negative organisms like Klebsiella spp
and E.coli. Neutralising Swabs are also sold separately.
Ideal For: Detecting Listeria spp.
including L. monocytogenes from food contact surfaces,
equipment or environmental. Routine Sampling for Pathogens is a
fundamental part of Good Manufacturing Practice (GMP)and essential for
adhering to a HACCP (Hazard Analysis Critical Control Points) plan. This
product can be used to compliment (or in conjunction with) the ISO11290-1
testing/detection methodology.
Known Limitations:
1. SwabSURE is intended as a screening test. All presumptive
positive results should be confirmed by standard or molecular bacteriological
methods.
2. It is recommended that after sampling food contact areas
are wiped. If used for direct food contact the item must be removed/discarded
after use (so not sold).
3. Has not been validated for uncleaned food surfaces. The
use of SwabSURE for these purposes may result in false positives or
spurious/unreadable results.
4. The absence of a colour change does not necessarily imply
the absence of target bacteria. Reasons for this may include:
a. Inadequate neutralization of sanitizing agents.
b. Numbers of target organisms are below the detection level
of the test.
c. The target organisms, although present may be severely
damaged and unable to be resuscitated.
5. The Incubation temperature can have an effect on false
positive rate. A temperature of 25°C is recommended and will result in fewer
false positive results. This is particularly important when sampling areas that
are difficult to maintain e.g. Drains, intricate equipment. These can act as
microbial reservoir increasing the false positives from organisms like Bacillus spp.
and Enterococcus spp. Please see alternative chromogenic DGS-L01
which uses a more specific colour change system. The occurrence of false
positive results is an important indicator of suspect cleaning and sanitizing
practices due to concentration of background flora and should flag
a review.
Shelf Life: Up to 12 months - Components should not be used after the expiry date printed on them or the carton label.
Storage: Store at 2-8°C when not in use. The
pre-moistened swabs should be stored at 4-25°C.
Sterilization Method: Terminally Sterilized
(detection media autoclaved, swab gamma irradiated)